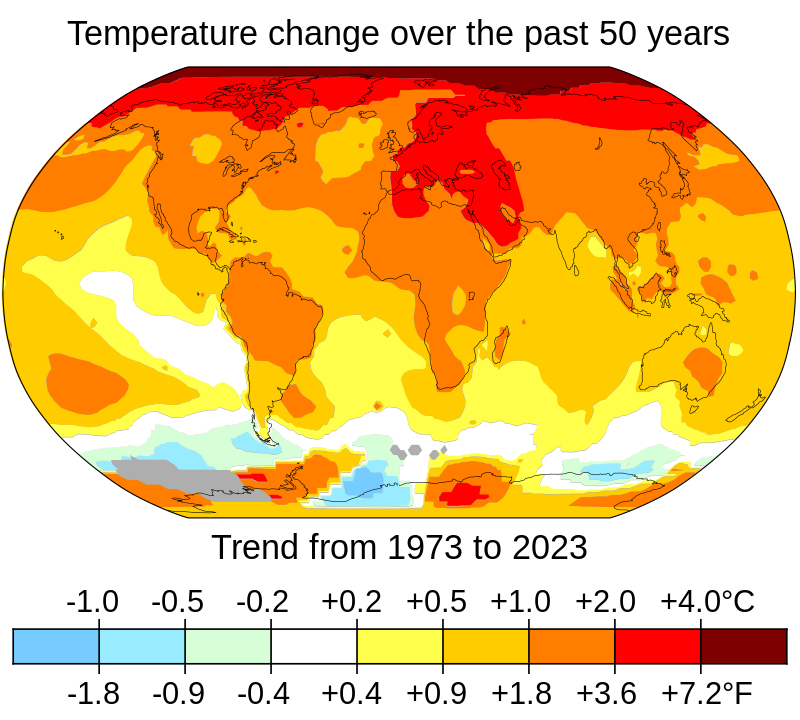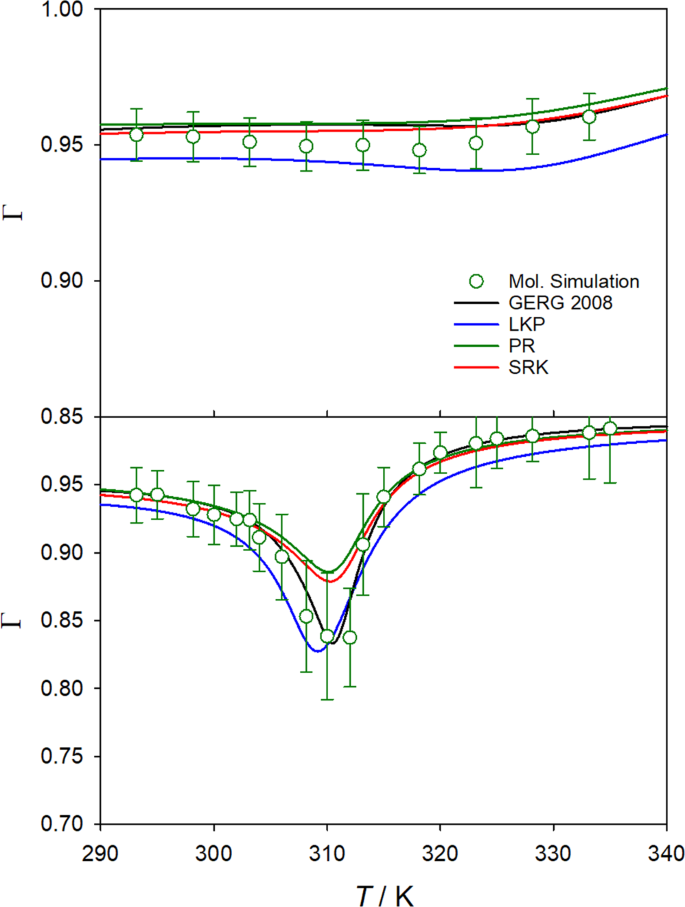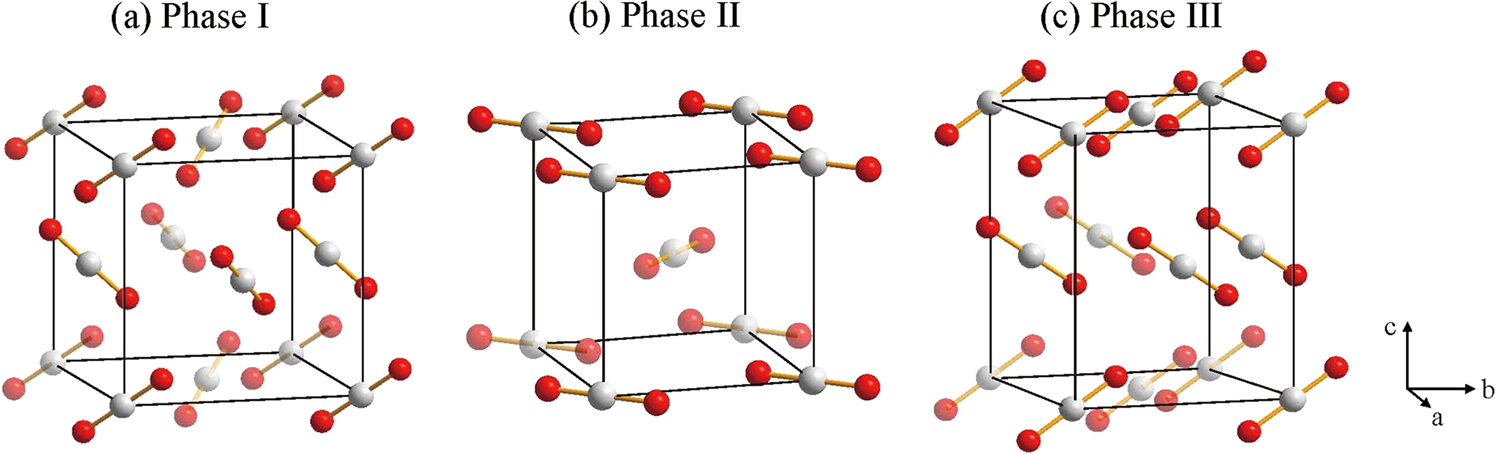
Predicting the phase diagram of solid carbon dioxide at high pressure from first principles | npj Quantum Materials

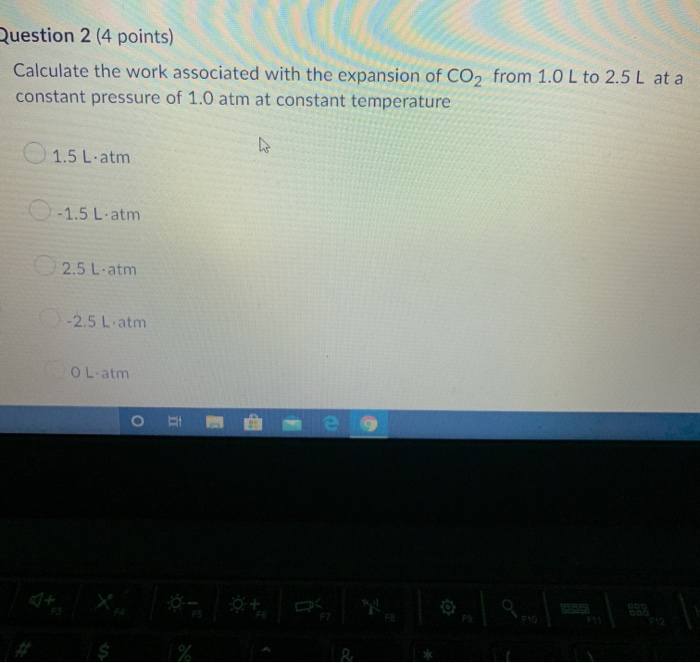
Chapter 2 Chem.docx - Ans: d Calculate the work for the expansion of CO2 from 1.0 to 5.8 liters against a pressure of 1.0 atm at constant | Course Hero

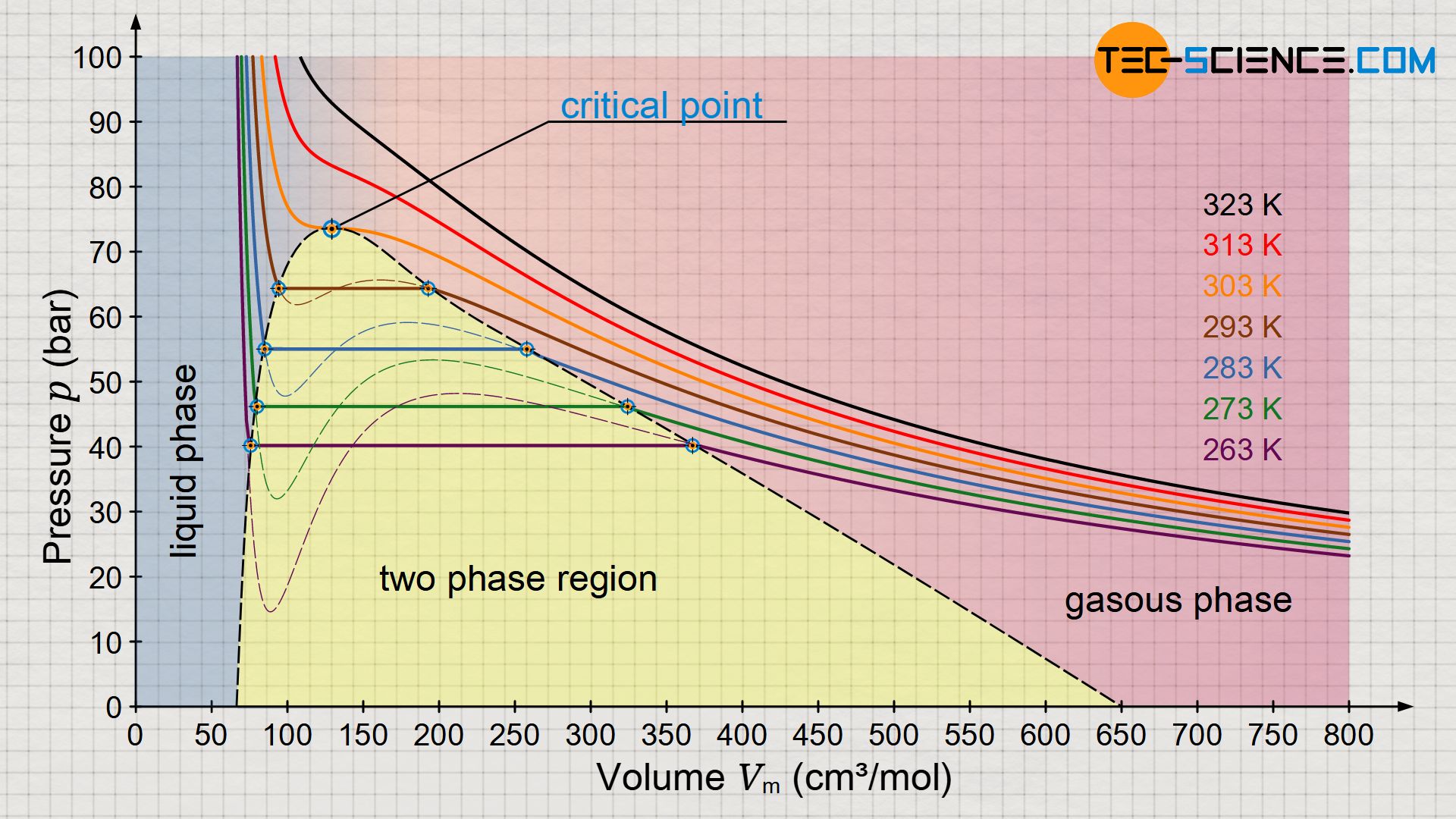
SOLVED: Problem Set#9 Thermochemistry Submitted You received no credit for this question in the previous att 2 Enter your answer in the provided box 393-g quantity of dry ice (solid carbon dioxide)

Solubility of Carbon Dioxide in Aqueous Solutions of Monoethanolamine in the Low and High Gas Loading Regions | Journal of Chemical & Engineering Data

Chapter 2 Chem.docx - Ans: d Calculate the work for the expansion of CO2 from 1.0 to 5.8 liters against a pressure of 1.0 atm at constant | Course Hero

Experimental Study on the Density-Driven Carbon Dioxide Convective Diffusion in Formation Water at Reservoir Conditions | ACS Omega
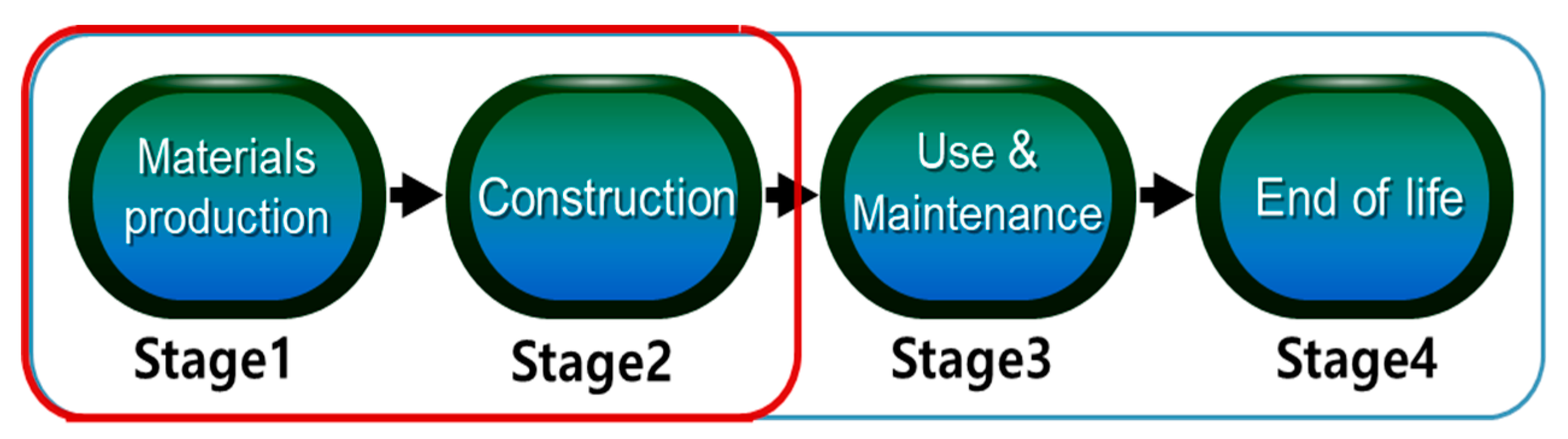
Applied Sciences | Free Full-Text | CO2 Emission Calculation Method during Construction Process for Developing BIM-Based Performance Evaluation System